General Program Questions
Data Collection
Data Use and Privacy
Educational Webinars
Eligibility
Implementation
GENERAL PROGRAM QUESTIONS
Back To Top
1. What are the benefits of participating?
The Agency for Healthcare Research and Quality (AHRQ) Safety Program for MRSA Prevention team will work closely with your surgical services team to enhance the approaches your hospital takes to optimize surgical site infection (SSI) prevention, with a focus on methicillin-resistant Staphylococcus aureus (MRSA) SSI prevention.
In addition to using evidence-based, practical implementation strategies, the program uses the Comprehensive Unit-Based Safety Program (CUSP) quality improvement method. The CUSP method will help you improve teamwork, communication, and leadership engagement to support a culture of patient safety. Together, these approaches will improve staff knowledge of infection prevention and help them identify approaches to successfully reduce and prevent SSI, with a focus on infections caused by MRSA.
By participating, your surgical service will—
- Reduce SSIs, with a focus on those caused by MRSA
- Promote and optimize infection prevention procedures
- Enhance teamwork and communication around MRSA prevention
- Improve patient safety and safety culture
Our team of subject matter experts has expertise in both SSI and MRSA prevention and CUSP and will be readily accessible for coaching, technical assistance, and ongoing education. Visit the Eligibility FAQ section or sign up for an informational webinar to find out if you qualify to participate in the program.
2. Who is sponsoring the program?
This program is funded and guided by AHRQ, which is part of the U.S. Department of Health and Human Services. Johns Hopkins Medicine’s Armstrong Institute for Patient Safety and Quality is conducting the work in collaboration with NORC at the University of Chicago.
3. What does it cost our hospital to participate in the program?
Participation in the program is free. Participating hospitals will not incur any fees to receive assistance as a part of this program and will not receive any payment for participation.
4. Will CME and CEU credits be awarded for participation?
CME and CEU credits will be available for participating physicians, physician assistants, and nursing staff.
5. What is the commitment for the program?
This program asks for an 18-month commitment. Orientation webinars to introduce the program and train representatives from your hospital surgical services team(s) about MRSA prevention will begin in January 2023. The following table shows surgical services team tasks and submission dates.
Task
|
Submission Date
|
|
Review and sign the Letter of Commitment for hospital participation (surgical services level) in the Safety Program.
|
January 2023
|
|
Assemble a multidisciplinary CUSP team for your surgical services team. The team could consist of a surgeon, nurse champion, and infection preventionist to lead and coordinate local efforts while participating in the AHRQ Safety Program and sustain those efforts after the program is completed. Each team also will identify a CUSP team lead and/or data coordinator who will facilitate data collection at your site.
|
November 2022 - December 2022
|
|
Ensure that members of your team have access to the Safety Program website. This website hosts all educational content associated with the Safety Program.
|
|
Submit quarterly infection prevention data for the 12 months prior to the start of the program (January 2022–December 2022). A team lead or data coordinator can submit data.
|
April 2023
|
|
Participate in educational programs, including an orientation webinar and monthly educational webinars. Team leaders are strongly encouraged to participate in the live webinars. Other clinicians and staff members are encouraged to join live webinars, or at a minimum, access and engage with content (e.g., recorded presentations; audio presentations; educational materials for staff, clinicians, and patients) on the program website.
|
January 2023 – June 2024
|
|
Meet regularly as a team (e.g., monthly) to discuss the content of the webinars, review current infection prevention practices, and identify areas for improvement in your practice.
|
|
Submit quarterly infection prevention data. A team lead or data coordinator can submit data.
|
6. What is required of our hospital to participate in the program?
Each surgical service team will identify a CUSP team, including a team lead. The CUSP team lead must be either a surgeon or a nursing champion with the surgeon’s endorsement and support. Other CUSP team members may include anesthesiologists, anesthesia team, nurses, nurse practitioners, nurse educators, physician assistants, infection preventionists, environmental services personnel, medical assistants, surgical technicians, pain control consultive services, first assistants, unit clerks/administrative assistants, and other members of the hospital staff affiliated with the participating surgical services. Each surgical services team will also identify a team member who will facilitate data collection at your site. Often this is the Infection Control Department’s data analyst. Activities include the following—
- Participate in regular webinars (30–60 minutes each). Participants will receive training via webinars held once or twice per month.
- Determine and implement approaches to promote and improve infection prevention. Using approaches discussed in the webinars, surgical services teams will develop policies and procedures that utilize program strategies including chlorhexidine gluconate (CHG) treatment (bathing), decolonization, MRSA surveillance, antimicrobial prophylaxis, improvement of hand antisepsis and hand hygiene, and implementation of other evidence-based infection prevention bundles.
- Collect and review data. Your team is encouraged to work together to review quarterly electronic health record and surgery registry data extracts of infection prevention team-level data, as well as the program provided quarterly benchmark reports that summarize deidentified data from similar teams.
7. How much time does this program take?
We anticipate that the CUSP team leads and clinical staff on the CUSP Team will likely need to spend a minimum of 3 hours per month on the AHRQ Safety Program for MRSA Prevention. Approximately 1 hour will be devoted to participating in webinars. The remaining hours will be spent implementing the measures discussed in the webinars or participating in office hours. Time will also be necessary to collect, submit, and review data.
8. When is the deadline to enroll?
The deadline to enroll in the Surgical Services program is February 28, 2023. We encourage you to submit an application to join the program as soon as possible as space in the program will be limited.
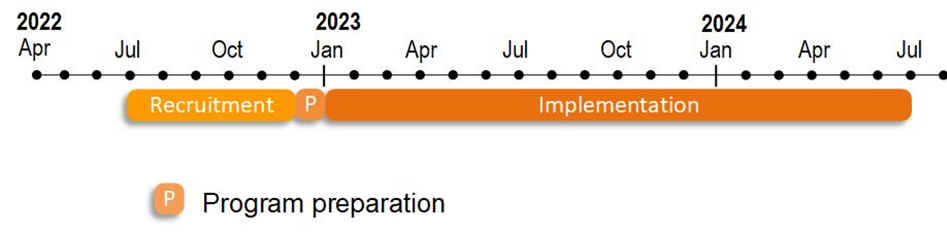
9. What is the timeline for the program?
Preparation for the Surgical Services program will begin in December 2022, and program implementation will begin in January 2023 (January 2023 through June 2024).
10. Is a contract required to participate in the program?
No, a contract is not required to participate in the program. To apply for the program, your surgical services team lead or other primary contact will need to complete the online application form available on the Join the Program page.
The program requests a Letter of Commitment signed by a hospital executive/administrator, surgeon, and clinician champion. This is not a contract; rather, it is an agreement to work with the Safety Program. We ask for these signatures to ensure support for the program from the hospital’s leadership and to approve the minimum time commitment of 3 hours per month per surgical services team. However, the actual program commitments/requirements will be fulfilled by the surgical service team of participating staff.
11. We already employ evidence-based MRSA prevention tactics. How can joining this program improve our MRSA infection rates and contribute to greater sustained adoption?
In addition to using evidence-based strategies, the program uses the Comprehensive Unit-based Safety Program (CUSP) quality improvement method, which promotes communication, teamwork, and leadership engagement to support a culture of patient safety, developing a greater sense of teamwork within surgical services teams.
CUSP engages and empowers staff. Participants in the program will learn how to address attitudes, beliefs, and culture issues that often pose challenges to implementing and improving adherence to the implementation of evidence-based strategies. The CUSP program accesses the wisdom of the frontline staff to help teams root out and define patient safety problems in the surgical services team and to develop and test strategies to solve these problems.
The program will provide tools such as auditing tools, checklists, and guides for performing mini root-cause analyses for healthcare-associated infections to use throughout the program. Between the audit tools and reports from the program and your Infection Preventionist, you will be better able to track your progress, understand where your implementation barriers are, and develop and carryout plans to overcome those barriers. The development and continued monthly work of the CUSP team and the appropriate and scheduled use of the auditing tools will help participating surgical services teams sustain their SSI prevention work and use the above techniques to begin to approach other patient safety issues in the team.
12. Is a data use agreement (DUA) required to participate in the program?
No, a DUA is not required for participation in the program. The data are being collected both for program assessment purposes and as a tool for each site’s own quality improvement efforts. Each surgical services team should use the data it collects to evaluate its infection prevention efforts. NORC at the University of Chicago and Johns Hopkins Armstrong Institute for Patient Safety and Quality will use the collected data to assess the adoption and effectiveness of the program overall. Additionally, each surgical services team can compare its team-level data for SSI events to similar teams.
Since the program only requests deidentified information and does not request any protected health information (PHI), program participation does not require a data use agreement. However, if your hospital determines that it still needs a DUA to define the transaction, we can provide a template for your review.
13. What material do I need to complete before the program begins?
You need to complete the online application and a Letter of Commitment. The application requests general hospital and surgical services-level demographic information, as well as contact information. The Letter of Commitment should be signed by a hospital executive/administrator, a clinician champion, and a surgeon.
Johns Hopkins Armstrong Institute for Patient Safety and Quality has determined this program is not human subjects research (Johns Hopkins IRB #IRB00284760). This program does not involve human subjects research, so no institutional review board (IRB) approval is required. Your hospital should be able to frame this program as a quality improvement study. If your hospital requires an IRB submission for informational purposes, we will be glad to help you as needed.
DATA COLLECTION
Back To Top
14. What data are being collected?
Please see the tables below.
Data Collection Tools
Tool
|
Purpose
|
To Be Completed by
|
Frequency of Data Collection
|
|
Hospital Survey on Patient Safety Culture (HSOPS)
|
To collect information on patient safety issues
|
CUSP team members
|
Twice - at the beginning and the end of the intervention period (months 1 and 18)
|
|
Infrastructure Assessment (Gap Analysis)
|
To evaluate existing resources and processes and identify areas of improvement to facilitate interventions to reduce the incidence and prevalence of surgical site infections (SSIs), especially SSIs caused by methicillin-resistant Staphylococcus aureus (MRSA).
|
Split between Team Lead and Infection Preventionist
|
Twice - at the beginning and the end of the intervention period (months 1 and 18)
|
|
Implementation Assessment (Team Checkup Tool)
|
Checklist of key actions to guide staff members towards a culture of safety by using guidelines, tools, and resources.
|
One staff member per surgical services team
|
Monthly
|
To evaluate the changes in infection prevention during the 18-month program, your surgical services CUSP team lead or data coordinator will be asked to submit the clinical outcomes data outlined in the tables below.
Retrospective monthly data for the past 12 months prior to implementation (January 2022 through December 2022) should be pulled once. Electronic data pulls of monthly data should be pulled on a quarterly basis from January 2023 through June 2024.
We estimate the initial data pull will take 60 minutes, and subsequent quarterly data pulls will take 30 minutes each.
The program team will offer flexibility around data collection dates to minimize barriers to program participation. The program team will work with your surgical services team to determine a data collection schedule that will work best for you.
Clinical Data Extracts
Neurosurgical and Orthopedic Surgical Services Teams